
Interference pattern: the pattern you would get if the electrons were waves,Įach wave passing through both slits at once and then interfering But what you actually see on the detector screen is an Then you would expect them to pile up around two straight lines behind the There's a second one which can detect where the electrons that made it In it electrons (or other particles like photons or neutrons) are fired oneĪt a time all over a screen containing two slits. One of the most famous demonstrations of wave-particle duality
#SCHRODINGER PICTURE PLUS#
Scattering off a crystal confirmed the wave-like nature of electrons (see the Plus article Quantum uncertainty). In the late 1920s experiments involving particles Sprung from theoretical considerations inspired by Einstein's theory of relativity. In some situations and like waves in others.ĭe Broglie's idea, which he announced in the 1920s, wasn't based on experimental evidence, rather it The tinyīuilding blocks of matter, such as electrons, also behave like particles Suffered from the so-called wave-particle duality. Inspired by the schizophrenic behaviour of light theīroglie took a dramatic step in this journey: he postulated that not only light, but also matter To unify the physics of optics (which was all about waves) and mechanics (which was all about particles). The double slit experiment: The top picture shows the interference pattern created by waves passing though the slits, the middle picture shows what you'd expect to see when particles are fired through the slits, and the bottom picture shows what actually happens when you fire particles such as electrons through the slits: you get the interference pattern you expect from waves, but the electrons are registered as arriving as particles.Įinstein's result linked in with the age-old endeavour, started in the 17th century by Christiaan Huygens and explored again in the 19th century by William Hamilton: Light was as waves and sometimes it was as particles," says The situation that sometimes the correct way of describing Physicist Max Planck who had already guessed this formula in 1900 in his work on black body radiation. Here is Planck's constant, an incredibly small number named after the The number of photons is proportional to the intensity of the light, and the energy E of each photon is proportional to its frequency f: You need to think of a beam of light as a stream of particles, whichĮinstein dubbed photons. Photoelectric effect (see the Plus article Light's identity crisis) Wave picture wasn't entirely correct either. Then, after the work of many scientists, including the theoretical understanding provided by James Clerk Maxwell, we discovered that light was "According to Newton, light was described by particles. His work on gravity, was also interested in optics," saysīouatta. Sometimes it is better to think of them as The problem is that the objects quantum mechanics tries to describeĭon't always behave like tiny little billiard balls. In quantum mechanics we ask the same question, but the answer is tricky because position and momentum are no longer the right variables to describe. "The kind of question we then ask is: if we know the initial conditions of a system, that is, we know the system at time what is the dynamical evolution of this system? And we use Newton’s second law for that.
#SCHRODINGER PICTURE FULL#
For example, if you’ve got a table full of moving billiard balls and you know the position and the momentum (that’s the mass times the velocity) of each ball at some time, then you know all there is to know about the system at that time : where everything is, where everything is going and how fast. "In classical mechanics we describe a state of a physical system using position and momentum," explains Nazim Bouatta, a theoretical physicist at the University of Cambridge.
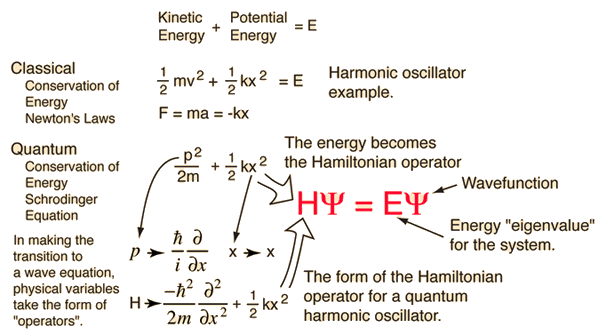

TheĬore equation of this theory, the analogue of Newton's second law, is called To describe this tiny world you need quantum mechanics,Ī theory developed at the beginning of the twentieth century. Smallest scales, for example electrons orbiting the nucleus of anĪtom, they realised that things get very weird indeed and that Newton's laws no longerĪpply. Or can it? When people first started considering the world at the Schrödinger's equation is named after Erwin Schrödinger, 1887-1961. Our basis ketsĪre time-independent but quantum states are built from them with time-dependent coefficients: The Schr ö dinger picture is the one we are used to. To d n ( t ) = e i E n ( 0 ) t ∕ ℏ c n ( t )), weĭid something which has a rather fancy name: we switched to the interaction picture. When at the start we, for convenience, absorbed the time dependence due to
